 Health & Physiology
Health & Physiology
Driving down malaria
Mosquitoes are the worlds’ deadliest animal due to their role in spreading Malaria. The revolutionary molecular technique known as CRISPR may help containing mosquitoes populations, preventing the disease to spread.
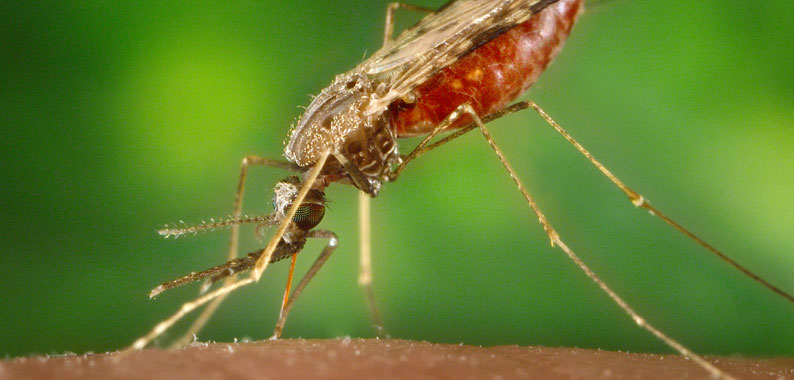
Mosquitoes are the deadliest animals on earth, having killed more people than wars and plagues combined. This is because they spread debilitating diseases like malaria - which affects more than 200 million people each year. Despite a momentous effort to combat the disease over the past two decades, we are unlikely to completely eliminate malaria without new interventions. Scientists have now turned to the tools of genetic engineering for new solutions.
Malaria is caused by a parasite, Plasmodium falciparum, that needs to develop in both a human and mosquito host to survive. If we make it impossible for the parasite to develop in the mosquito, then we could stop malaria dead in its tracks. Recent advances in genetic engineering have made this possible by introducing new traits into the mosquito that affect its own survival, or its ability to carry the parasite. The problem is no longer the process of engineering, but how to spread this modification throughout mosquitoes in the wild.
To explain the problem, we need to consider what happens to a genetic modification over time. Any modification that we make to a mosquito chromosome is inherited like any other gene. Just like us, mosquitoes contain two copies of every chromosome and each of their offspring inherit just one of the two copies. If a few modified mosquitoes were released into the wild, that modification would not spread because it only ever gets inherited by half of the offspring.
In 2003, Professor Austin Burt postulated an exciting method to overcome this problem using something called a "gene drive". Gene drives are genetic modifications that can spread through an entire population by biasing their own inheritance. This means that such modifications will over-ride the classical Mendelian inheritance laws, with a mechanism also called "Super-Mendelian". By increasing their chances of being passed on, they ensure that more and more mosquitoes will have gained the modification over time.
Drawing upon Burt's original theory, we used a pair of molecular scissors called "CRISPR" to engineer gene drives that work through a mechanism of copy and paste. A gene drive made with CRISPR was inserted into a specific region within one chromosome of the mosquito "Anophels gambiae" and programmed to cut its unmodified partner chromosome. When the broken chromosome is damaged it repairs as precisely as possible by using the modified partner chromosome as a template, but in doing so it unwittingly copies the gene drive inside. In this way, it doesn't matter which of the two chromosome copies are inherited because the gene drive is present on both (see figure "How gene drives spread"). Amazingly, we saw that our gene drive mosquitoes passed on the modification to more than 99% of their offspring!
In our lab, we designed three different gene drives that would make mosquitoes infertile. This sounds weird, because it shouldn't be possible to spread the gene drive if the mosquitoes are unable to produce offspring? We achieved this by programming our gene drives to affect only female mosquitoes and only when both of their parents were modified. Thus, the gene drive has no detrimental effect when only one of their parents is a carrier, allowing it to spread when gene drive mosquitoes mate to those in the wild (see figure "Using gene drives to eliminate malaria"). To test this hypothesis, mosquitoes carrying one of the three gene drive were released into a cage of unmodified mosquitoes and their reproduction was tracked over time. More and more mosquitoes were found to carry the modification with each generation, showing for the first time that a gene drive can be designed to invade natural population of dangerous insects.
If mosquitoes carrying such gene drive are released into the wild, it is expected that they could spread throughout an entire population within just 20-30 mosquito generations. Because mosquitoes reproduce very quickly, this could take just a couple of years starting from a relatively small release. As the gene drive gets into more and more mosquitoes, mating between two gene drive carriers will become more frequent, and result in the generation of female mosquitoes that are infertile. This will lead to a population crash and potentially reduce the number of biting mosquitoes below the critical threshold needed to support continued malaria transmission.
A critical problem for malaria control is delivering effective interventions to those who need it most. With gene drive, the mosquitoes do the hard work. This self-sustaining, species-specific and long-term intervention could be used to boost current control strategies like bed-nets, insecticides and vaccines. Gene drives may be the last intervention needed to tip the fight against malaria in our favour - we want to make malaria a disease of the past.
Original Article:
Hammond A, Galizi R, Kyrou K, Simoni A, Siniscalchi C, Katsanos D, Gribble M, Baker D, Marois E, Russell S, Burt A, Windbichler N, Crisanti A, Nolan T. A CRISPR-Cas9 gene drive system targeting female reproduction in the malaria mosquito vector Anopheles gambiae. Nature Biotechnology. 2015;34(1):78-83. doi:10.1038/nbt.3439Next read: How early-life adversity gets under the skin by Joanne Ryan
Edited by:
Massimo Caine , Founder and Director
We thought you might like
Fighting back antibiotic resistance: a new hope from the soil
Feb 24, 2016 in Microbiology | 4 min read by Dan KramerThe colour beige: heating up the fat
Apr 1, 2016 in Health & Physiology | 3.5 min read by Caterina Da RèOne run a day keeps the...cancer away!
May 5, 2016 in Health & Physiology | 3.5 min read by Per thor Straten , Manja IdornSERINC5: a blood cell guardian against HIV
May 13, 2016 in Health & Physiology | 3.5 min read by Massimo PizzatoMore from Health & Physiology
Tobacco smoking and other exposures shut off cancer-fighting genes
Aug 31, 2024 in Health & Physiology | 3 min read by Jüri Reimand , Nina AdlerA hidden clock that times cytoplasmic divisions
Aug 30, 2024 in Health & Physiology | 3 min read by Cindy OwWhen two kinases go for a dance
Aug 2, 2024 in Health & Physiology | 4 min read by Ioannis Galdadas , Francesco Luigi Gervasio , Pauline JuyouxAwakening the thymus to cure SARS-CoV-2 infection: a matter of genes
Jul 27, 2024 in Health & Physiology | 3.5 min read by Stefano Marullo , Cheynier RemiKeeping the balance: How epigenetics monitors cancer genes
May 13, 2024 in Health & Physiology | 4 min read by Zach Gray , Madison Honer , Johnathan WhetstineEditor's picks
Trending now
Popular topics


